This article gives a full summary of vaccine manufacturing technology in the past ten years. It talks about new immunization agent platforms, approved vaccines, better production technologies and stronger analysis and testing abilities.
I. Progress and Approvals in Vaccine Development
- Approval of New Platform Vaccines
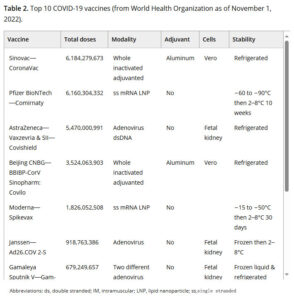
DNA and RNA vaccines, including viral vector and mRNA‐LNP vaccines, develop very fast. Since 2012, ten new viral vector vaccines and two mRNA‐LNP vaccines have been approved. These vaccines work against Japanese encephalitis, dengue fever, Ebola, SARS‐CoV‐2 and other diseases.
Some viral vector vaccines use special methods. One example is GamEvac‐Combi. It uses different vectors, like VSV and Ad5, to carry antigens.
For example, insect cell culture (Sf9 cells) with baculovirus expression systems is utilized for large-scale manufacturing of influenza immunization agent hemagglutinin (HA) protein.
Cell lines including Vero and PER.C6 can be adapted to suspension culture for viral propagation. Optimized CHO cell culture technologies (e.g., those described by Watterson et al. for SARS-CoV-2 vaccine candidates) are applicable to immunization agent manufacturing, including the production of truncated varicella-zoster virus glycoprotein E for the Shingrix immunizing agent.
During the COVID-19 pandemic, vaccines developed by the University of Oxford based on the replication-deficient adenoviral platform achieved successful large-scale amplification. Suspension cell culture was employed to express adenoviral vectors delivering the spike protein, supporting mass immunization agent supply.
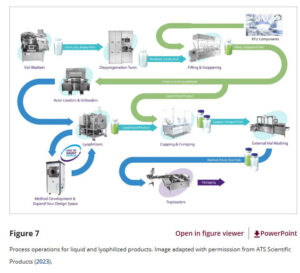
- Microbial Production
Traditional Applications and New Progress
Microbial fermentation platforms are employed to produce a wide range of vaccines. For example, Saccharomyces cerevisiae expresses the HPV L1 protein, which self-assembles into virus-like particles (VLPs) to form the Gardasil immunization agent. Hepatitis B virus surface antigen can be expressed by either Saccharomyces cerevisiae or Pichia pastoris. Pneumococcal conjugate vaccines are manufactured by conjugating antigens expressed by specific bacterial strains.
Novel culture platforms have made remarkable progress. The Dyadic platform, based on the fungus Thermothelomyces heterothallica, enables rapid development of stable strains, delivers high yields, and offers scalable and cost-effective production. Its COVID-19 immunizing agent candidate is currently under clinical evaluation. The automated platform based on Pichia pastoris developed at the Massachusetts Institute of Technology can produce diverse proteins, including immunization agent antigens, and is now applied to the production of rotavirus immunizing agent candidates.
The production of protein antigens faces challenges in exposing critical epitopes. In mRNA immunization agent manufacturing, Escherichia coli fermentation is the primary method for producing plasmid DNA (pDNA) templates. While cell-free synthesis of pDNA has advanced, its cost-effectiveness remains to be improved. Strain selection and sequence characteristics are critical considerations in pDNA production, and innovative approaches for conjugate immunization agent manufacturing have also emerged.
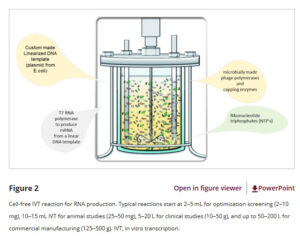
3.Equipment Innovation and Development Trends
Fermentation equipment has transitioned from stainless steel pressure vessel fermenters to single-use bioreactors. Single-use bioreactors offer multiple advantages, such as eliminating cleaning and sterilization procedures and improving production flexibility, yet their maximum practical scale is limited to approximately 3,000 liters by process conditions.
Single-use options have been established for purification workflows, covering from depth filtration to chromatographic columns. Immunizing agent manufacturers are striving to build a supply chain ecosystem for single-use systems, and the industry is jointly promoting sustainable recycling and circular economy strategies for single-use components.
III. Conclusion and Outlook
The past decade has witnessed remarkable progress in immunization agent process technology, including the development of novel platforms, the approval of diversified vaccines, innovations in production technologies, and enhanced analytical and characterization capabilities. These advances are of great significance in preventing infectious diseases and developing future therapeutic cancer vaccines. Moving forward, the immunization agent research and development field will continue to integrate cutting-edge technologies, methodologies and platforms to promote global human health.
Original Citation: B. Buckland, G. Sanyal, T. Ranheiim, et al. immunizing agent process technology—A decade of progress. Biotechnology and Bioengineering, 2024.
About Ferbio
Ferbio boasts extensive expertise in the manufacturing of various bioreactors and pressure vessels. We have assembled a professional team covering the fields of bioreaction, fermentation technology, mechanical engineering and automated control. Our scientific research and process technologies maintain a leading position domestically and reach world-class standards internationally, providing you with comfortable, reliable and reassuring product experiences.
